GNLM – Myanmar Daily News, Myanmar Latest News, Myanmar News Today
Digital directories set for 17 Mandalay Region pagodas
According to the Mandalay Region government, digital directories are currently being installed at 17 renowned pagodas across the Mandalay Region. […]
YGEA to open 32nd gold training course for free on 2 May
The Yangon Gold Entrepreneur Association (YGEA) will open the 32nd gold training course for free on 2 May at the […]
YGEA to open 32nd gold training course for free on 2 May
The Yangon Gold Entrepreneur Association (YGEA) will open the 32nd gold training course for free on 2 May at the […]
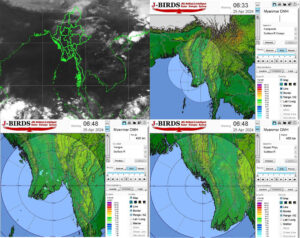
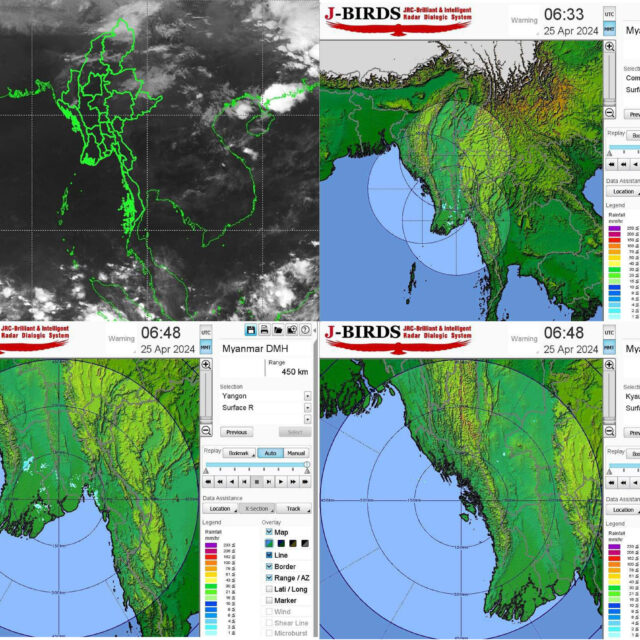
Central Myanmar, Delta expect slight rise in temperatures later this month
In central Myanmar and the delta region, temperature is forecasted to rise slightly by about one degree on 28-29 April, […]
Investors Club’s 31st Anniversary held in New Delhi
The Ministry of Foreign Affairs (MoFA) stated that U Moe Kyaw Aung, Myanmar’s Ambassador to India, participated in the 31st […]
Myanmar celebrates World Immunization Week 2024
Investors Club’s 31st Anniversary held in New Delhi
Central Committee on Copyright meeting 1/2024 held
No Use
By Kyaw Myint Tun (Paris) For ease of reference, let’s continue to refer to these hypocrites, who descend from their […]
Why Not Read Novels?
By Hu Wo (Cuckoo’s Song) According to Oxford, novels are stories long enough to fill a complete book, in which […]
Journey Through the Timeless Tea Legacy
By Yin Nwe Ko Tucked away in a heritage shophouse on Mosque Street in Singapore’s Chinatown, Pek Sin Choon is […]
Sustain health of mother earth with plastic waste management
T he Earth serves as a vital habitat for all forms of life, encompassing humans, animals, plants, and various objects. […]
Shakespeare’s Enduring Legacy
S hakespeare was a famous writer. He wrote many good and winning plays. He created many attractive plays. When I […]